 In a pair of patent owner victories, the U.S. Court of Appeals for the Federal Circuit issued decisions limiting the applicability of obviousness-type double patenting — known as OTDP — to invalidate or limit the term of earlier issued, but later expiring patents. On December 7, the Federal Circuit issued two opinions that address the scope and applicability of OTDP: Novartis Pharmaceuticals v. Breckenridge Pharmaceutical and Novartis AG v. Ezra Ventures. Although these decisions are both important in their own right, they are also significant because they limit the reach of two Federal Circuit cases that district courts have used to apply OTDP more broadly to invalidate patents that had later expirations: Gilead Sciences v. Natco Pharma and AbbVie v. Mathilda & Terence Kennedy Institute of Rheumatology Trust. Breckenridge and Ezra limit OTDP’s tread on patent terms extended pursuant to Congressionally approved programs.
In a pair of patent owner victories, the U.S. Court of Appeals for the Federal Circuit issued decisions limiting the applicability of obviousness-type double patenting — known as OTDP — to invalidate or limit the term of earlier issued, but later expiring patents. On December 7, the Federal Circuit issued two opinions that address the scope and applicability of OTDP: Novartis Pharmaceuticals v. Breckenridge Pharmaceutical and Novartis AG v. Ezra Ventures. Although these decisions are both important in their own right, they are also significant because they limit the reach of two Federal Circuit cases that district courts have used to apply OTDP more broadly to invalidate patents that had later expirations: Gilead Sciences v. Natco Pharma and AbbVie v. Mathilda & Terence Kennedy Institute of Rheumatology Trust. Breckenridge and Ezra limit OTDP’s tread on patent terms extended pursuant to Congressionally approved programs.
Patentees may look to the reasoning of Breckenridge and Ezra for support when defending against OTDP arguments. The cases emphasize that OTDP — a judicially — created doctrine designed to prevent patent owners from extending the term of a patent by claiming obvious variants in different patents with different expiration dates—and double patenting generally, are designed to prevent gamesmanship in prosecution that would unjustifiably extend patent terms beyond their statutory limits. OTDP is not intended to be, and should not be applied as, a mechanical doctrine to reduce patent terms that are otherwise statutorily proper.
Breckenridge and Patent Expiration Dates
In Breckenridge, the Federal Circuit considered the application of OTDP to patents that had different expiration dates based whether the application leading to the patent was filed before or after June 8, 1995, when the Uruguay Round Amendments Act went into effect. Under URAA, patents issuing from applications filed before June 8, 1995 (pre-URAA), were entitled to a term that is the longer of 20 years from the earliest effective filing date for priority, or 17 years from the patent issue date. Applications filed after June 8, 1995 (post-URAA), receive terms of 20 years from their earliest effective filing date. As a result, later-filed applications in a patent family may expire before earlier-filed applications, as was the case in Breckenridge. The patent application and issuance timeline of Breckenridge is illustrated in the court’s diagram below.
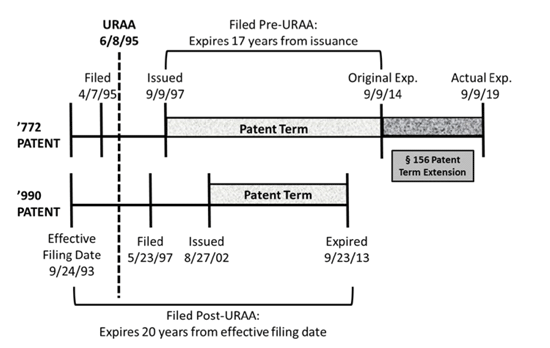
The Federal Circuit held that the earlier expiring post-URAA ’990 patent could not serve as a double-patenting reference against the later expiring ’772 patent.
In reaching this conclusion, the Federal Circuit first limited Gilead — and, in turn, AbbVie — to the narrow factual situations of those cases. In Breckenridge, the Federal Circuit observed that Gilead’s use of expiration dates to determine if one patent can be an OTDP reference to another did not control because Gilead was limited to the situations where both the challenged patent and the reference patent issued from post-URAA applications. Further, the Federal Circuit observed that cases like Gilead, in which the target and reference patents have different effective filing dates and hence different expiration dates post-URAA, can give rise to gamesmanship whereby a patentee claims obvious variants in unrelated applications resulting in a longer term, but that in a case like Breckenridge, where the effective filing dates were the same, there was little if any risk of gamesmanship.
The Federal Circuit also reasoned that in enacting the URAA transition statute, Congress intended that patentees should receive the full benefit of the pre-URAA patent term without being limited by post-URAA term rules. Applying that reasoning, the Federal Circuit found that, unlike Gilead where the expiration date of the patents was used to determine whether the earlier expiring patent could be an invalidating reference, in Breckenridge the ’772 patent’s issue date was the proper “reference point” for the double patenting analysis. Therefore, because the ’990 patent did not issue until after the pre-URAA ’772 patent, the ’990 patent could not serve as a double-patenting reference against the ’772 patent.
Ezra and Patent Term Extensions
In Ezra, the Federal Circuit considered OTDP’s effect on a valid patent-term extension (PTE) pursuant to 35 U.S.C. § 156. As the court noted, Section 156 was enacted as part of the Hatch-Waxman Act “to restore the value of the patent term that a patent owner loses during the early years of the patent because the product cannot be commercially marketed without approval from a regulatory agency,” such as the U.S. Food and Drug Administration. The relevant timeline for Ezra is illustrated in the court’s chart reproduced below, which shows that the ’229 patent would have expired before the later issued ’565 patent, but for the patent term extension.
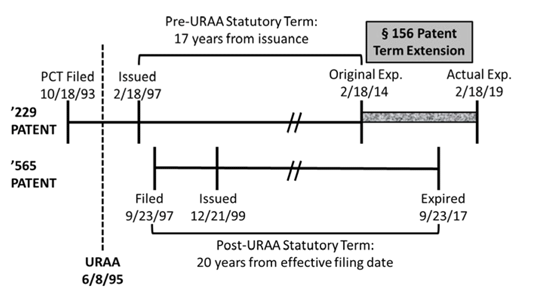
In this case, Ezra — the patent challenger — raised two primary arguments:
- That the ’229 patent’s PTE “effectively” extended the terms of both the ’229 and ’565 patent; and
- The ’229 patent is invalid for OTDP because the PTE causes it to “expire” after the ’565 patent.
The Federal Circuit rejected both arguments. As to the first argument, the Federal Circuit observed that in enacting Section 156, Congress allowed the extension of the term of a single patent for up to five years, but left the choice of which patent to extend to the patent owner. The court found “no reason to read ‘effectively’ as a modifier to ‘extend’ in the language of § 156(c)(4).” The Federal Circuit rejected the second argument based on what it called a “logical extension” of its previous decision in Merck & Co. v. Hi-Tech Pharmacal Co., which held that terminally disclaimed patents may receive PTE extending beyond the term of a reference patent. The Federal Circuit noted that the statutory language (“shall”) of Section 156 mandated that the patent term be extended if the other requirements for a PTE were met. Thus, the court held that “if a patent, under its pre-PTE expiration date, is valid under all other provisions of the law, then it is entitled to the full term of its PTE.”
Responding to OTDP Arguments
For both pre- and post-URAA patents, practitioners should consider Breckenridge and Ezra when drafting and responding to OTDP arguments. This judicially-created doctrine was designed to protect against gamesmanship during prosecution which could result in unjustifiably extended patent terms, nothing further. For pre-URAA patents, the date of issuance is now a relevant consideration, expiration dates alone do not control the analysis of what may count as a reference patent. For post-URAA patents, the Federal Circuit has made clear that the appearance or risk of gamesmanship during prosecution may be an important factor in whether the double patenting doctrine should be applied to limit a patent’s term. Patent owners and practitioners will still also need to consider the question unanswered by these cases when, evaluating priority claims and seeking any changes to a patent term other than PTE, such as patent term adjustments (PTA). Although neither Breckenridge nor Ezra speak directly to the issue, factual situations where a patent’s term extends beyond the expiry of a potential reference patent, whether by PTA or otherwise, may or may not be insulated from OTDP challenges.
Takeaways
OTDP remains an important issue, particularly for patents in the life sciences. Understanding OTDP and tracking changes in the law are important because patentees often end up with related patents with claims of different scope (e.g., genus and species, compounds and methods of treatment, etc.). Breckenridge and Ezra should give patent owners some comfort when facing decisions regarding what patent to apply for a PTE. Courts now recognize that this choice was left to the patentee and that PTE alone will not invalidate its patents for OTDP.
Going forward, Breckenridge and Ezra will also be important considerations in many OTDP arguments. Breckenridge extensively distinguished Gilead and AbbVie, essentially limiting both to their facts: post-URAA patents with different priority dates. Ezra furthers this distinguishing concept making clear that the problematic gamesmanship in Gilead is not present when patents issue from applications that ultimately claim priority to the same application. Breckenridge and Ezra thus suggest that double-patenting will be a more challenging argument against related patents, which — by definition — claim priority to the same application.
Following Merck and Ezra, it seems clear that PTE alone is not vulnerable to OTDP. PTE, along with “additional” non-PTE term, however, may be vulnerable to OTDP. At the district court level, the Breckenridge case suggested that PTE plus some additional patent term extending beyond the expiration date of a reference patent may be vulnerable to double-patenting. (See Novartis Pharmaceuticals v. Breckenridge Pharmaceutical) Breckenridge did not address this issue as the case was resolved on the pre- and post-URAA patent status.